Miconazole Nitrate CAS 22832-87-7 Assay 98.0%-102.0% Antifungal High Purity
Shanghai Ruifu Chemical Co., Ltd. is the leading manufacturer of Miconazole Nitrate (CAS: 22832-87-7) with high quality. Ruifu Chemical has been supplying APIs (active pharmaceutical ingredients) and pharmaceutical intermediates more than 15 years. Ruifu Chemical can provide competitive price, excellent service.
Purchase Miconazole Nitrate, please contact us by e-mail: alvin@ruifuchem.com
| Chemical Name | Miconazole Nitrate |
| Synonyms | (±)-Miconazole Nitrate; (±)-Miconazole Nitrate Salt; 1-[2-(2,4-Dichlorobenzyloxy)-2-(2,4-Dichlorophenyl)ethyl]-1H-Imidazole Nitrate; 1-(2,4-Dichloro-β-[(2,4-Dichlorobenzyl)oxy]phenethyl)imidazole |
| Stock Status | In Stock, Production Capacity 100 Tons/Year |
| CAS Number | 22832-87-7 |
| Molecular Formula | C18H14Cl4N2O·HNO3 |
| Molecular Weight | 479.14 g/mol |
| Melting Point | 178.0-184.0℃ |
| Storage Temperature | Store Long-Term in a Cool and Dry Place |
| Shipping Conditions | Ambient |
| COA & MSDS | Available |
| Origin of Product | Shanghai, China |
| Product Categories |
API (Active Pharmaceutical Ingredient) |
| Caution | Research Use Only and Are Not Intended for Human Use |
| Brand | Ruifu Chemical |
| Items | Specifications | Results |
| Appearance | White to Almost White Powder | Conforms |
| Identification | IR: Similar to Standard | Complies |
| Appearance of Solution | The Solution is Clear and Not More Intensely Coloured Than Reference Solution Y7 |
Complies |
| Solubility | Very Slightly Soluble in Water, Slightly Soluble in Methanol, Slightly Soluble in Alcohol |
Complies |
| Specific Optical Rotation | -0.10° - +0.10° (C=10 mg/ml in Methanol) | Complies |
| Related Impurities | ||
| Any Other Single Impurity | ≤0.25% | 0.16% |
| Total impurities | ≤0.50% | 0.39% |
| Melting Point | 178.0-184.0℃ | 180.0-181.0℃ |
| Loss on Drying | ≤0.50% (105℃ for 2 h) | 0.10% |
| Residue on Ignition | ≤0.20% | 0.09% |
| Assay / Analysis Method | 98.0%-102.0% | 99.5% |
| Particle Size | 100% less than 10um | Complies |
| Conclusion | The product has been tested and complies with the given specifications | |
Package: Bottle, Aluminium foil bag, 25kg/cardboard drum, or according to customer's requirement.
Storage Condition: Keep the container tightly closed. Store in a cool, dry and well-ventilated warehouse away from incompatible substances. Keep away from sunshine; avoid fire and heat sources; avoid moisture.
Shipping: Deliver to worldwide by air, by FedEx / DHL Express. Provide fast and reliable delivery.
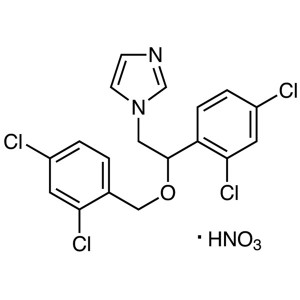
Miconazole Nitrate
C18H14Cl4N2O·HNO3 479.14
1H-Imidazole, 1-[2-(2,4-dichlorophenyl)-2-[(2,4-dichlorophenyl)methoxy]ethyl]-, mononitrate;
1-[2,4-Dichloro-β-[(2,4-dichlorobenzyl)oxy]phenethyl]imidazole mononitrate [22832-87-7]
DEFINITION
Miconazole Nitrate contains NLT 98.0% and NMT 102.0% of Miconazole Nitrate (C18H14Cl4N2O·HNO3), calculated on the dried basis.
IDENTIFICATION
Change to read:
• A. SPECTROSCOPIC IDENTIFICATION TESTS <197>, Infrared Spectroscopy: 197A or 197K (CN 1-May-2020)
• B. The retention time of the major peak of the Sample solution corresponds to that of the Standard solution, as obtained in the Assay.
ASSAY
• PROCEDURE
Solution A: Methanol, water, and 1 M triethylammonium acetate (30:70:1)
Solution B: Acetonitrile, methanol, and 1 M triethylammonium acetate (25:75:0.2)
Mobile phase: See Table 1.
Table 1
Time (min) Solution A (%) Solution B (%)
0 70 30
5 70 30
10 44 56
27 44 56
30 25 75
35 25 75
36 70 30
40 70 30
Diluent: Methanol and water (70:30)
System suitability solution: 0.1 mg/mL of USP Miconazole Nitrate RS and 6 µg/mL of USP Miconazole Related Compound F RS in Diluent. Sonication may be needed to aid dissolution.
Standard solution: 0.1 mg/mL of USP Miconazole Nitrate RS in Diluent. Sonication may be needed to aid dissolution.
Sample solution: 0.1 mg/mL of Miconazole Nitrate in Diluent. Sonication may be needed to aid dissolution.
Chromatographic system
(See Chromatography <621>, System Suitability.)
Mode: LC
Detector: UV 215 nm
Column: 4.6-mm × 10-cm; 2.6-µm packing L11
Column temperature: 40°
Flow rate: 0.8 mL/min
Injection volume: 10 µL
System suitability
Samples: System suitability solution and Standard solution
[NOTE-The relative retention times for miconazole related compound F and miconazole are 0.96 and 1.0, respectively.]
Suitability requirements
Resolution: NLT 1.5 between miconazole related compound F and miconazole, System suitability solution
Tailing factor: NMT 2.0, Standard solution
Relative standard deviation: NMT 0.73%, Standard solution
Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of miconazole nitrate (C18H14Cl4N2O·HNO3) in the portion of Miconazole Nitrate taken:
Result = (r /r ) × (C /C ) × 100
r = peak response of miconazole from the Sample solution
r = peak response of miconazole from the Standard solution
C = concentration of USP Miconazole Nitrate RS in the Standard solution (mg/mL)
C = concentration of Miconazole Nitrate in the Sample solution (mg/mL)
Acceptance criteria: 98.0%-102.0% on the dried basis
IMPURITIES
• RESIDUE ON IGNITION <281>: NMT 0.2%
• ORGANIC IMPURITIES
Solution A, Solution B, Mobile phase, Diluent, and Chromatographic system: Proceed as directed in the Assay.
Standard solution: 1.2 µg/mL each of USP Miconazole Nitrate RS, USP Econazole Nitrate RS, USP Miconazole Related Compound C
RS, USP Miconazole Related Compound F RS, and USP Miconazole Related Compound I RS in Diluent
Sample solution: 600 µg/mL of Miconazole Nitrate in Diluent. Sonication may be needed to aid dissolution.
System suitability
Sample: Standard solution
[NOTE-See Table 2 for relative retention times.]
Suitability requirements
Resolution: NLT 1.5 between miconazole related compound C and miconazole related compound I; NLT 1.5 between miconazole
related compound I and econazole; and NLT 1.5 between miconazole related compound F and miconazole
Relative standard deviation: NMT 3.0% for miconazole
Analysis
Samples: Standard solution and Sample solution
Calculate the percentage of miconazole related compound C, miconazole related compound F, miconazole related compound I, or
econazole nitrate in the portion of Miconazole Nitrate taken:
Result = (rU /rS ) × (CS /CU ) × 100
rU = peak response of miconazole related compound C, miconazole related compound F, miconazole related compound I, or
econazole nitrate from the Sample solution
rS = peak response of miconazole related compound C, miconazole related compound F, miconazole related compound I, or
econazole nitrate from the Standard solution
CS = concentration of USP Miconazole Related Compound C RS, USP Miconazole Related Compound F RS, USP Miconazole
Related Compound I RS, or USP Econazole Nitrate RS in the Standard solution (µg/mL)
CU = concentration of Miconazole Nitrate in the Sample solution (µg/mL)
Calculate the percentage of each specied and any individual unspecied impurity in the portion of Miconazole Nitrate taken:
Result = (rU /rS ) × (CS /CU ) × 100
rU = peak response of each specied and any individual unspecied impurity from the Sample solution
rS = peak response of miconazole from the Standard solution
CS = concentration of USP Miconazole Nitrate RS in the Standard solution (µg/mL)
CU = concentration of Miconazole Nitrate in the Sample solution (µg/mL)
Acceptance criteria: See Table 2.
Table 2
Name Relative Retention Time Acceptance Criteria, NMT (%)
Deschlorobenzyl econazole a 0.22 0.25
Miconazole quarternary salt b 0.57 0.25
Miconazole benzyl analog c 0.65 0.25
Miconazole related compound C 0.74 0.25
Miconazole related compound I 0.76 0.25
Econazole nitrate 0.78 0.25
Miconazole 2,6-isomer d 0.87 0.25
Miconazole 2,5-isomer e 0.94 0.25
Miconazole related compound F 0.96 0.25
Miconazole 1.0 -
Any individual unspecied impurity - 0.10
Total impurities - 0.5
a 1-(2,4-Dichlorophenyl)-2-(1H-imidazol-1-yl)ethanol.
b 2-(3-{2-[(2,4-Dichlorobenzyl)oxy]-2-(2,4-dichlorophenyl)ethyl}-1H-imidazol-3-ium-1-yl)-2-methylpropanoate.
c 1-[2-(Benzyloxy)-2-(2,4-dichlorophenyl)ethyl]-1H-imidazole.
d 1-{2-[(2,6-Dichlorobenzyl)oxy]-2-(2,4-dichlorophenyl)ethyl}-1H-imidazole.
e 1-{2-[(2,5-Dichlorobenzyl)oxy]-2-(2,4-dichlorophenyl)ethyl}-1H-imidazole.
SPECIFIC TESTS
Change to read:
• OPTICAL ROTATION <781A>, Procedures, Angular Rotation (RB 1-MAY-2019)
Sample solution: 10 mg/mL of Miconazole Nitrate in methanol
Acceptance criteria: -0.10° to +0.10° at 20° (RB 1-May-2019)
• LOSS ON DRYING <731>
Analysis: Dry at 105° for 2 h.
Acceptance criteria: NMT 0.5%
ADDITIONAL REQUIREMENTS
• PACKAGING AND STORAGE: Preserve in well-closed containers, protected from light. Store at controlled room temperature.
• USP REFERENCE STANDARDS <11>
USP Econazole Nitrate RS
USP Miconazole Nitrate RS
USP Miconazole Related Compound C RS
2-[(2,4-Dichlorobenzyl)oxy]-2-(2,4-dichlorophenyl)ethan-1-amine hydrochloride.
C15H13Cl4NO·HCl 401.53
USP Miconazole Related Compound F RS
1-{2-[(3,4-Dichlorobenzyl)oxy]-2-(2,4-dichlorophenyl)ethyl}-1H-imidazole.
C18H14Cl4N2O 416.13
USP Miconazole Related Compound I RS
1-{2-[(2-Chlorobenzyl)oxy]-2-(2,4-dichlorophenyl)ethyl}-1H-imidazole mononitrate.
C18H15Cl3N2O·HNO3 444.69
How to Purchase? Please contact Dr. Alvin Huang: sales@ruifuchem.com or alvin@ruifuchem.com
15 Years Experience? We have more than 15 years of experience in the manufacture and export of a wide range of high quality pharmaceutical intermediates or fine chemicals.
Main Markets? Sell to domestic market, North America, Europe, India, Korea, Japanese, Australia, etc.
Advantages? Superior quality, affordable price, professional services and technical support, fast delivery.
Quality Assurance? Strict quality control system. Professional equipment for analysis include NMR, LC-MS, GC, HPLC, ICP-MS, UV, IR, OR, K.F, ROI, LOD, MP, Clarity, Solubility, Microbial limit test, etc.
Samples? Most products provide free samples for quality evaluation, shipping cost should be paid by customers.
Factory Audit? Factory audit welcome. Please make an appointment in advance.
MOQ? No MOQ. Small order is acceptable.
Delivery Time? If within stock, three days delivery guaranteed.
Transportation? By Express (FedEx, DHL), by Air, by Sea.
Documents? After sales service: COA, MOA, ROS, MSDS, etc. can be provided.
Custom Synthesis? Can provide custom synthesis services to best fit your research needs.
Payment Terms? Proforma invoice will be sent first after confirmation of order, enclosed our bank information. Payment by T/T (Telex Transfer), PayPal, Western Union, etc.
| Hazard Symbols | Xn - Harmful |
| Risk Codes | R22 - Harmful if swallowed R43 - May cause sensitization by skin contact |
| Safety Description | 36/37 - Wear suitable protective clothing and gloves. |
| UN IDs | 2933199090 |
| WGK Germany | 3 |
| RTECS | NI4771000 |
| HS Code | 2933290090 |
Miconazole Nitrate (CAS: 22832-87-7) is a broad-spectrum antifungal drug commonly used to treat fungal infections. Miconazole Nitrate is a highly effective, safe and broad-spectrum antifungal agent that inhibits and kills dermatophytes, candida, yeast and other algae, ascomycetes, cryptococci, etc., and also has Gram-positive cocci and bacilli. Antibacterial effect. Mainly used for skin fungal infections, such as hands, feet, body, femoral hernia.
Intravenous Administration
1. Miconazole Nitrate (CAS: 22832-87-7) can be used for serious infections caused by Candida fungi, including peritonitis, pneumonia and urinary tract infections.
2. Miconazole Nitrate can also be used for chronic extensive mucocutaneous candidiasis;
Partial External Use
Miconazole Nitrate can be used for the treatment of cutaneous candidiasis, candida vulvovaginitis, body lice, femoral hernia, athlete's foot, tinea versicolor and so on.